Reconditioning of Adulterated Food Products
Course details
Self-Paced eLearning
69.00
45 Min
Thomas Hammack
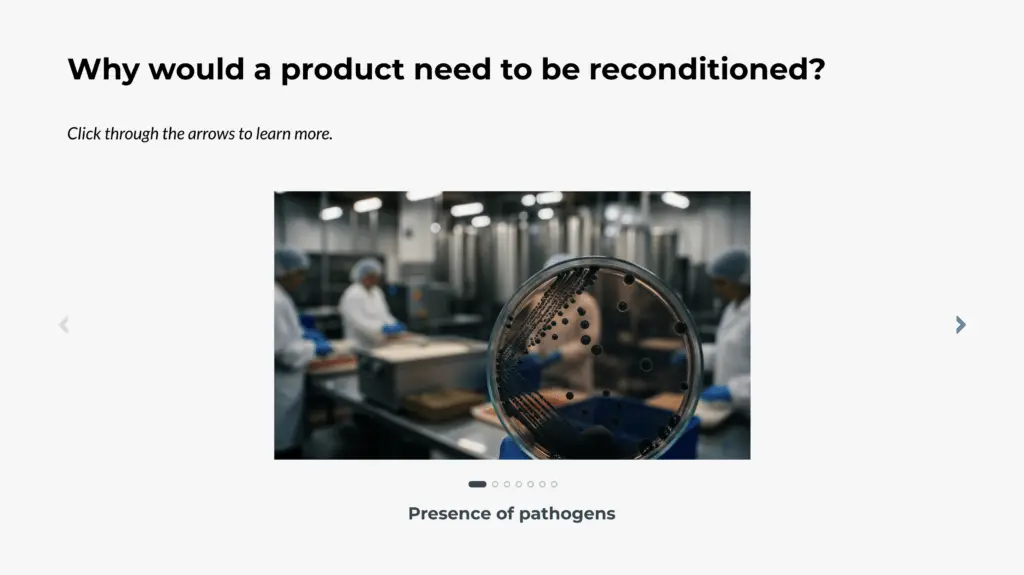
This course explains the regulatory and scientific considerations for reconditioning adulterated food products. When a product is determined to be adulterated, options may include recall, detention, or destruction unless reconditioning can be demonstrated to restore the product to a safe and compliant condition.
The course outlines when reconditioning is allowed, when it is prohibited, and how to develop a defensible reconditioning proposal for FDA review. Topics include applicable regulatory requirements, documentation expectations, and considerations for both domestic and imported products.
Emphasis is placed on demonstrating that reconditioned products meet safety standards for human or animal consumption and on aligning corrective actions with FDA expectations.
Benefits and Learning Objectives
After completing this course, participants will be able to:
- Define adulteration and identify conditions under which reconditioning may be considered
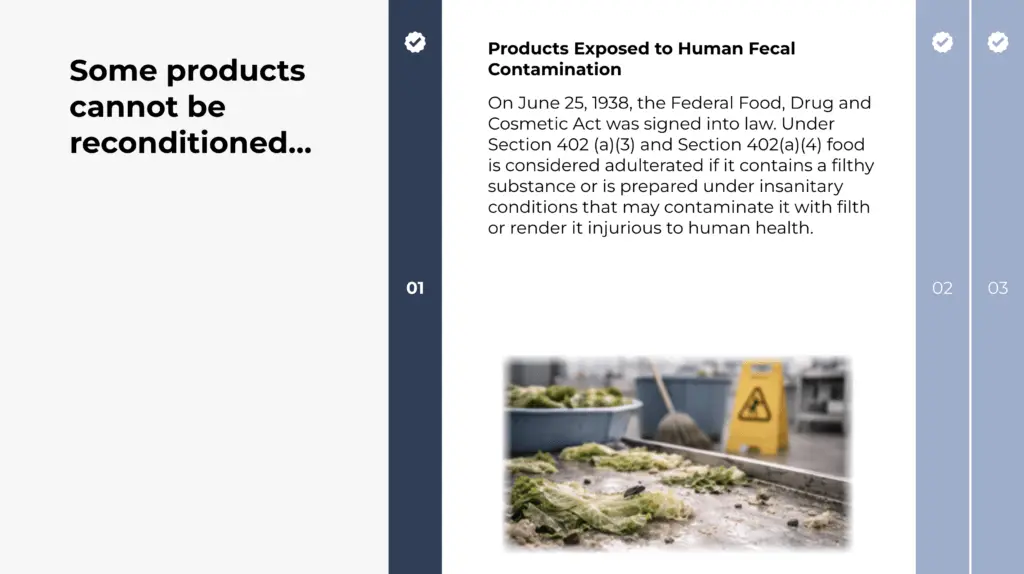
- Distinguish situations where reconditioning is permitted versus prohibited
- Describe FDA expectations for reconditioning proposals and supporting documentation
- Develop approaches to demonstrate that a product can be made safe for consumption
- Understand regulatory considerations for both domestic and imported products
- Evaluate corrective actions to ensure compliance and reduce regulatory risk

Agenda
- Definition and regulatory basis of adulteration
- Overview of reconditioning as a corrective action
- When reconditioning is permitted and when it is not allowed
- FDA requirements and expectations for reconditioning proposals
- Documentation and validation considerations
- Reconditioning of domestic versus imported products
- Demonstrating product safety post-reconditioning
Who Should Attend?
- Quality Assurance and Quality Control professionals
- Regulatory affairs and compliance teams
- Food safety and risk management personnel
- Operations and production managers
- Importers, suppliers, and distribution teams
Registration and Payment
To register for this course, please visit our training portal.

-
Vice President, Technical Services
IEH Laboratories & Consulting Group
Meet the Instructor
Thomas Hammack
Thomas Hammack joined IEH as Vice President of Technical Services in 2024. He has over 30 years of experience in the field of food microbiology, working at the FDA’s Center for Food Safety and Applied Nutrition (CFSAN). Starting in 1990, he began his tenure at the FDA as a research microbiologist until he became the Chief of the Microbial Methods Development Branch (MMDB) in 2009. In this role, he led a team of 25 microbiologists in developing and validating methods for the detection and characterization of bacterial pathogens and toxins in foods and on environmental surfaces. After a decade as Chief of MMDB, he concluded his tenure as a Senior Policy Analyst for Microbiology in CFSAN’s Office of Regulatory Science.
He is a co-author of FDA’s Bacteriological Analytical Manual’s (BAM) Salmonella, Cronobacter, and Food Sampling and Preparation of Sample Homogenate chapters. During his career, he Chaired the FDA’s Microbiology Methods Validation Subcommittee (MMVS) from 2017 to 2021, which oversees the validation of the microbiological methods used in the FDA’s Foods Program. He also chaired the FDA’s Bacteriological Analytical Manual (BAM) Council from 2009 to 2016. He remained a member of both the BAM Council and MMVS until his retirement at the end of 2023.
Thomas Hammack’s expertise extends beyond his roles at the FDA. He currently serves as an Expert Review Panel member for AOAC International, overseeing the validation of numerous methods for bacterial pathogens, including Salmonella, Listeria, and E. coli O157:H7. Since 2014, he has been a dedicated member of the MicroVal Technical Review Committee. Notably, he served as the Chair of the US Technical Advisory Group to ISO TC 34/SC 9 from 2011 to 2023, the committee responsible for the development of all ISO food microbiological methods.
Related Trainings
SELF-PACED ELEARNING
1.5 HOURS


