Developing Food Product Specifications
Course details
Self-Paced eLearning
99.00
1 Hour
Dr. Suf Alkhaldi
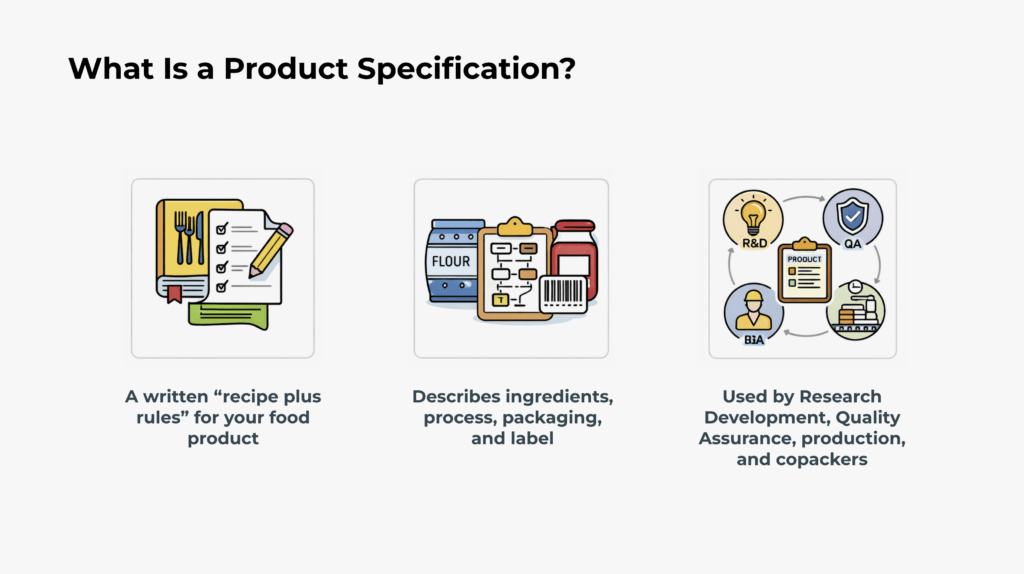
This course covers how to develop, review, and maintain food product specifications that support food safety, accurate labeling, and regulatory compliance. Product specifications function as the central reference point for how a product is defined, produced, and controlled across operations.
The course addresses key elements including product identity, ingredient and allergen controls, labeling requirements, and the distinction between Ready-to-Eat (RTE) and Cook-Before-Eating (CBE) foods. It also outlines how specifications support science-based safety limits, shelf life determinations, packaging requirements, and alignment with food safety plans. By the end of the course, participants will be able to create and manage clear, controlled, and compliant product specifications that protect consumers and their company.
Benefits and Learning Objectives
After completing this course, participants will be able to:
- Define the role of product specifications across R&D, Quality, Production, and co-manufacturing
- Explain how specifications support FDA and USDA compliance and reduce food safety and labeling risks
- Distinguish between Ready-to-Eat (RTE) and Cook-Before-Eating (CBE) foods and apply appropriate controls
- Identify and document ingredients, allergens, and labeling requirements accurately
- Apply science-based limits for processing, storage, and shelf life
- Maintain controlled specifications through change control and document management practices

Agenda
- Role and structure of food product specifications
- Regulatory considerations and compliance requirements
- Product identity, ingredients, and allergen controls
- RTE vs. Cook-Before-Eating classification
- Critical limits, shelf life, and storage conditions
- Packaging and labeling requirements
- Document control and change management
Who Should Attend?
- Quality Assurance and Quality Control professionals
- Food safety and regulatory compliance teams
- R&D and product development staff
- Operations and production personnel
- Co-manufacturers and suppliers responsible for product specifications
Registration and Payment
To register for this course, please visit our training portal.

-
Senior Vice President, Technical Services
IEH Laboratories & Consulting Group
Meet the Instructor
Dr. Suf Alkhaldi
Dr. Suf Alkhaldi joined IEH as the Senior Vice President of Technical Services in 2023. He earned his M.S. and Ph.D. in Microbiology and Cell Molecular Biology from Oklahoma State University. He also conducted a postdoctoral study in Rumen Microbiology in Animal and Dairy Science at the University of Georgia and served as the manager of the DNA Microarray Lab at Yerkes National Primate Research Center at Emory University.
Before joining IEH, Dr. Alkhaldi spent over two decades at FDA which has been characterized by his exceptional contributions to innovation and regulatory excellence. He began as a researcher and food outbreak investigator at the Center for Food and Applied Nutrition (CFSAN), focusing on developing cutting-edge methods for identifying pathogenic bacteria in food using molecular techniques. With over 29 peer-reviewed manuscripts, 14 reviewed papers and book chapters, and co-editing “The Bad Bug Book,” he demonstrated a strong commitment to knowledge-sharing and food safety. Dr. Alkhaldi has played an instrumental role in resolving the FDA’s 2008 pepper outbreak investigation.
Throughout his extensive career at FDA, Dr. Alkhaldi held various roles, including Project Manager, Supervisor, and Senior Science Advisor in the Office of the Chief Scientist. He actively participated in influential working groups and the FDA Funded Centers of Excellence, reinforcing his dedication to advancing microbiology and FDA drug policies. Adding to his accomplishments, Dr. Alkhaldi received over 20 FDA awards between 2000 and 2023.
Dr. Alkhaldi was selected to lead the Office of Safety within the Office of Regulatory Affairs (ORA). His exceptional leadership during the COVID-19 pandemic was evident as he led a team of 24 Industrial Hygienists and safety professionals. Their dedication ensured the safety of 5,000 FDA employees and provided support for 16 FDA laboratories, demonstrating his significant impact in fostering a safe and innovative regulatory environment within the FDA and the broader field of food microbiology.
Related Trainings
SELF-PACED ELEARNING
2 HOURS

