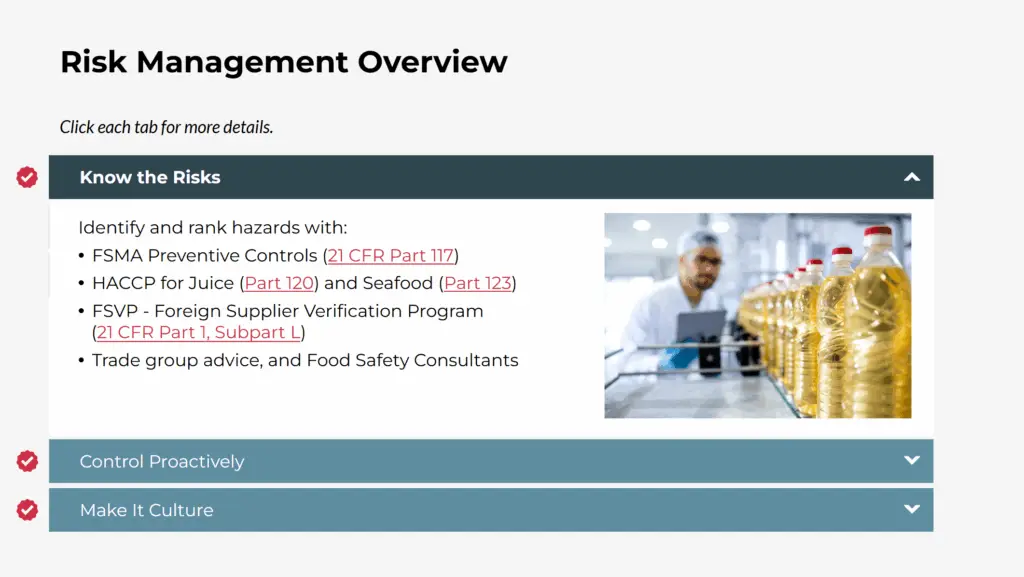
This course provides a practical framework for developing Quality Assurance and Quality Control (QA/QC) systems that align with FDA requirements, including FSMA, 21 CFR 117, and HACCP. Participants will learn how to prepare for FDA audits, reduce the risk of recalls and 483s, and build defensible SOPs, accurate records, and meaningful metrics that protect both food safety and brand integrity.
Through expert instruction, case studies, quizzes, and downloadable templates, learners will gain the skills to turn regulatory expectations into daily practices that support compliance and long-term business success.


To register for this course, please visit our training portal.

Dr. Suf Alkhaldi joined IEH as the Senior Vice President of Technical Services in 2023. He earned his M.S. and Ph.D. in Microbiology and Cell Molecular Biology from Oklahoma State University. He also conducted a postdoctoral study in Rumen Microbiology in Animal and Dairy Science at the University of Georgia and served as the manager of the DNA Microarray Lab at Yerkes National Primate Research Center at Emory University.
Before joining IEH, Dr. Alkhaldi spent over two decades at FDA which has been characterized by his exceptional contributions to innovation and regulatory excellence. He began as a researcher and food outbreak investigator at the Center for Food and Applied Nutrition (CFSAN), focusing on developing cutting-edge methods for identifying pathogenic bacteria in food using molecular techniques. With over 29 peer-reviewed manuscripts, 14 reviewed papers and book chapters, and co-editing “The Bad Bug Book,” he demonstrated a strong commitment to knowledge-sharing and food safety. Dr. Alkhaldi has played an instrumental role in resolving the FDA’s 2008 pepper outbreak investigation.
Throughout his extensive career at FDA, Dr. Alkhaldi held various roles, including Project Manager, Supervisor, and Senior Science Advisor in the Office of the Chief Scientist. He actively participated in influential working groups and the FDA Funded Centers of Excellence, reinforcing his dedication to advancing microbiology and FDA drug policies. Adding to his accomplishments, Dr. Alkhaldi received over 20 FDA awards between 2000 and 2023.
Dr. Alkhaldi was selected to lead the Office of Safety within the Office of Regulatory Affairs (ORA). His exceptional leadership during the COVID-19 pandemic was evident as he led a team of 24 Industrial Hygienists and safety professionals. Their dedication ensured the safety of 5,000 FDA employees and provided support for 16 FDA laboratories, demonstrating his significant impact in fostering a safe and innovative regulatory environment within the FDA and the broader field of food microbiology.
We use cookies to improve your experience on our site. By using our site, you consent to cookies.
Manage your cookie preferences below:
Essential cookies enable basic functions and are necessary for the proper function of the website.
These cookies are needed for adding comments on this website.
Google reCAPTCHA helps protect websites from spam and abuse by verifying user interactions through challenges.
Google Tag Manager simplifies the management of marketing tags on your website without code changes.
Statistics cookies collect information anonymously. This information helps us understand how visitors use our website.
Google Analytics is a powerful tool that tracks and analyzes website traffic for informed marketing decisions.
Service URL: policies.google.com (opens in a new window)
You can find more information in our Privacy Policy and .